Mary Davis was 35 years old when she got breast cancer.
“Most of the time when people get it when they’re young, it’s more aggressive. So she went through a year and half of extremely aggressive chemotherapy, including all the way to what’s called a stem cell transplant,” says her husband Mark Davis, a professor of chemical engineering at the California Institute of Technology. “It was just horrible.”
All of her hair fell out. The treatment permanently damaged her hearing. For the stem cell therapy, the mother of three had to be in isolation for weeks at the hospital, away from her young children. Like many cancer patients, Mary Davis wondered if the treatment would kill her rather than the disease.
Unfortunately, Davis’s experience wasn’t unusual. One of the reasons that chemotherapy is so devastating is that cancer drugs can kill any cell in the body, not just the tumor cells, says Andrew Tsourkas, an associate professor of bioengineering at the University of Pennsylvania. “One of the main issues for current chemotherapies is that they distribute nonspecifically throughout the body, and you end up with these adverse effects because you’re hitting targets that you’d rather avoid,” he says. With chemotherapy, doctors can’t control where the drug goes, so healthy cells get the same dose as tumor sites. “You get a lot of off-target toxicity.”
“As a surgeon who treats cancers at the late stages,” says Dr. B. Mark Evers, a colon cancer specialist and director of the Markey Cancer Center at the University of Kentucky, “I can cure [patients] if it hasn’t spread past the colonic wall. But if it’s metastasized to the liver, then their chances of cure are pretty nonexistent, even with the best of chemotherapies.” Though that might change, he says, if you could deliver high concentrations of the drugs into the tumors themselves and limit the toxicity in other parts of the body. “That becomes a very good treatment agent.”
While the Davises were sitting in the hospital more than 20 years ago, they were thinking there had to be a better way. At one point, Mary turned to her husband, who at the time specialized in chemical engineering for the petrochemical industry, and said, “You guys should start working on this at Caltech,” Mark recalls.
Which is exactly what he did. Davis and his team came up with a way to deliver more chemotherapy drugs to cancer cells and less to healthy ones: nanoparticles. It was a very new concept when they started in the 1990s—in fact, they first called them “colloids.” Since then, proposed uses for nanoparticles in medicine have multiplied, ranging from cardiovascular disease to vaccines. But nowhere is their promise greater than in improving cancer treatments.
The Rise of the Small
Cancer is not a small problem. The American Cancer Society estimates that in 2013 nearly 1.7 million Americans will get cancer and over 580,000 people will die of the disease. It’s the second most common cause of death in this country after heart disease. If the cancer metastasizes, or spreads to other parts of the body, the outlook can be grim.
When Davis’s wife was sick, it was understandably an emotional time for him, one he dealt with in a manner befitting a scientist. “Basically for the month that I was sitting in the hospital there with her—where she was suffering quite a bit—I was sitting there reading as much as I could,” he says. Not novels or magazines, but scientific literature about cancer treatment. “And eventually,” Davis says, “we came into this idea of what we’re doing now.”
Now, Davis and many other researchers are using nanotechnology as a cancer treatment. In general, nanotechnology refers to the use of materials on very small scales. In the field of cancer treatments, that lies between 1 and 100 nanometers, according to the National Cancer Institute. For scale, an antibody is about 10 nm, or 1/100,000,000,000 of a meter, and a flu virus is about 100 nm.
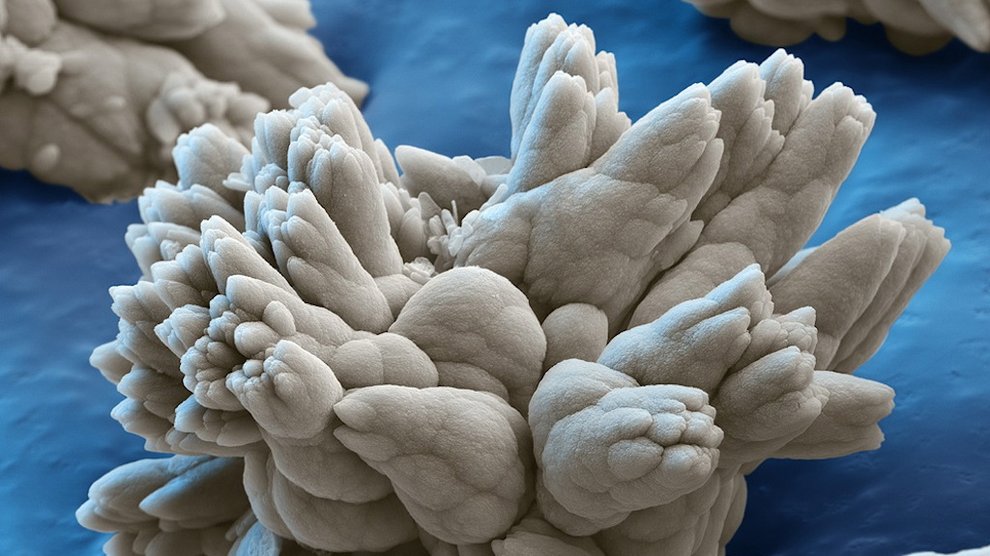
Nanotechnology has come a long way in the past few decades. In the early days, scientists used to mix chemicals together and hope some useful particles came out. Now, they can make virtually any size or shape of nanoparticle out of a wide variety of materials. Furthermore, it’s now much easier to make hollow nanoparticles, in which scientists can stash various chemicals and compounds, including cancer drugs. Nanoparticles can team up with highly toxic cancer drugs and deliver most of the poison right where it needs to go—into malignant tumors.
The strategy takes advantage of the way that cancer cells grow. Blood vessels in healthy tissues are narrow, blocking the passage of anything larger than about 2 to 6 nanometers. But cancerous tumors grow pell-mell fast, resulting in broken and permeable blood vessels, open to objects in the hundreds of nanometers. A 50-nanometer nanoparticle would be largely barred from healthy tissues, but it would have no trouble slipping into a tumor.
Depending on what nanoparticles are made of, they can also take advantage of the body’s special chemistry. Many are coated with a polymeric layer of polyethylene glycol, known as PEG, a material that prevents the nanoparticle from being attacked by the body’s immune system. “It doesn’t necessarily evade it, but it helps disguise the particle so it gets less readily recognized. So it extends its circulation time in the body,” Tsourkas says. The longer the medicine stays in the body, the higher the chance it will reach a tumor and exact beneficial damage.
Dozens of nanomedicines are in clinical trials right now, according to Tsourkas, and a few have been approved by the FDA. One of these is the medicine Doxil, which contains hollow nanoparticles with the cancer drug doxorubicin inside. It’s primarily used to treat ovarian cancer and multiple myeloma, and it was one of the first nanoparticle-based cancer drugs to be approved by the FDA.
One of Davis’s drugs is showing potential, too. It is currently in phase II of four phases of clinical trials. It’s a self-assembling, long-chain molecule based on sugars that’s linked to the cancer drug camptothecin. The particle, about 25 to 30 nanometers in diameter, is smaller than 100-nanometer Doxil. Its narrow profile is one of its main advantages, Davis says. Doxil can’t penetrate as deeply into the tumor, meaning its active ingredient, camptothecin, is only released on the periphery and has farther to travel to reach the core of the tumor. Davis’s drug, with its smaller particle size, can “move though the tumor and go inside of the tumor cells and then give the drugs off,” he says.
Of the hundreds of patients in the phase II trial of Davis’s drug, many have had a good response. And in a way, they have his wife to thank for it. “If she wouldn’t have encouraged us to do this, we wouldn’t have probably ever gotten started,” he says.
To Target or Not To Target
Despite their successes, nanoparticle-based drugs are not perfect. The differential between healthy cells killed and tumor cells killed “is not as great as you’d like it to be,” Tsourkas says. Nanomedicines are more specific than traditional cancer medicines—which act wherever they fall—but still there are times when not enough of the drug makes it to the tumor sites.
Fortunately, we know how to make targeted nanomedicines; chemists can attach molecules that target cancer cells to the outside of nanoparticles. Inside, the chemotherapy drug molecules are carried unaltered until they hit the tumor site. “What targeting potentially allows you to do is get a larger percentage of the drug you’re administering to the patient to the diseased tissue, while hopefully reducing how much gets to the healthy tissue,” Tsourkas says.
However, both Tsourkas and Davis say that opinions are mixed on whether targeting is actually needed. Adding a targeting molecule adds a layer of complexity, which makes the drug more expensive to manufacture and also more prone to errors. “When you add targeting, it significantly increases the expense because now you have to also create the targeting ligand, you have to create the nanoparticles, and then you have to have a simple and efficient way to combine the two,” Tsourkas says. “The cost of going down this road can be extremely high.”
Producing targeted nanoparticles in large enough quantities for clinical trials is also problematic. “Any time you make a drug that will be injected into a patient, you need to make sure you’re injecting the same thing every time,” Tsourkas says. To achieve that consistency, scientists have to simplify. Adding additional features, like a targeting molecule, makes it harder to ensure the formula is the same every time, he says. “I’m sure that’s going to be a roadblock for a lot of nanoparticle drug formulations.”
“If you look at the whole history, there’s probably less than ten examples of people making a nanoformulation with a targeting agent that’s actually gone into a clinical trial,” Davis says. His targeted drug, which is currently in clinical trials, is one of the few to have made it that far.
Still Far to Go
Davis’s wife Mary eventually got better, but there are hundreds of thousands of people each year who don’t. “There are a bunch of challenges, obviously,” Tsourkas says. “We have our work cut out for us.”
Davis agrees it’s a long road. “I have to be honest with you, when we got started, when we were about somewhere between a year one and year two, we hadn’t…I almost quit. We weren’t making any progress at all,” he says, recalling his frustration at the time. “But finally we started to make some progress, and things moved along quite well from then.”
Things are moving along well for the entire field, too. Tsourkas says that there are many promising applications of nanomedicines capturing the scientists’ attention, especially in developing vaccines and other methods of stimulating the immune system to fight disease.
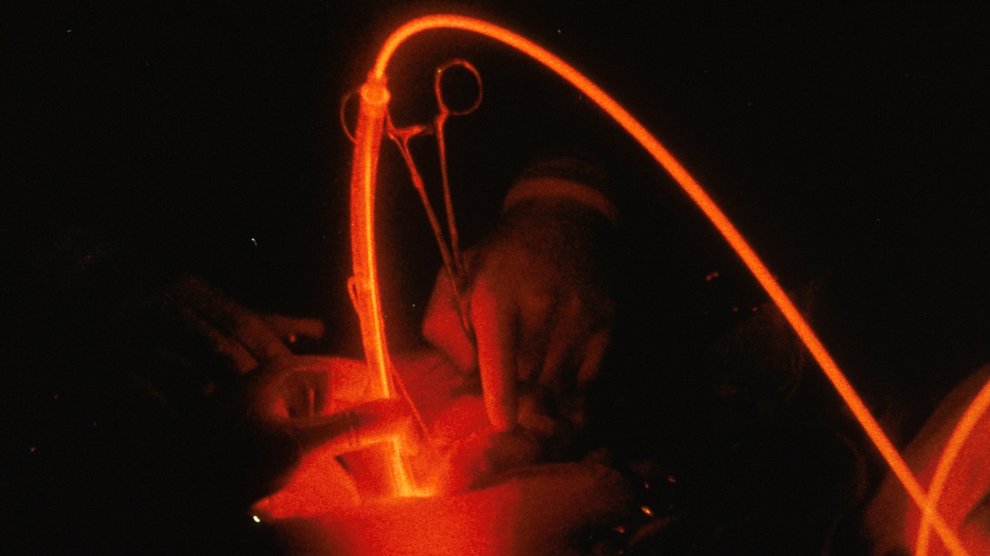
There are also several groups using nanoparticles with photodynamic therapy, a technique that destroys tumor tissue by changing a drug’s chemistry with the energy from an external light source, typically a red laser. By only shining the light where the tumor is, doctors can limit damage to healthy cells. Specially-prepared nanoparticles can also help monitor progress by absorbing the light and glowing back in a different color. Using a light-detecting instrument, doctors and researchers can see where the drug is and if it is working.
Davis’s success hasn’t slowed him down. Currently, he is working on different targeting system in which antibodies help nanoparticles pinpoint cancer cells. With this new particle, he says, they can deliver more of the active ingredient and control more precisely how quickly it is released compared with current leading cancer drugs.
Both scientists and doctors are hopeful these technologies will become potent allies in our battle against cancer. Nanomedicines should fulfill that promise “in the not too distant future,” says Evers, the director of the Markey Cancer Center. “I would say in the next five years, they are going to be, hopefully, fairly commonplace.”

